The evolution of sex and recombination is one of the most puzzling biological questions. Despite the wide phylogenetic distribution of sexual reproduction in eukaryotes, sex and recombination are evolutionarily costly processes: recombination (i.e., crossover and gene conversion) re-shuffles the parental genotypes that have successfully survived to reproduction, bearing risks of creating low-fitness or inviable offspring genotypes. In contrast, asexuals are rare and usually occupy the tips on the tree of life, suggesting their evolutionarily short persistence.
An important approach to understand why sex and recombination have been maintained is to examine asexual genomic evolution. Although obligate asexuality has evolved independently in multiple eukaryotic lineages, the molecular mechanisms leading to loss of meiosis and establishment of asexuality remain poorly understood. For example, despite a large proportion of asexuals have a hybrid origin, it remains unclear why hybridization leads to meiosis suppression. Due to the loss of meiosis that involves multiple DNA repair pathways (e.g., DNA mismatch repair), DNA repair in asexuals is most likely impaired and results in distinct mutation spectrum compared to sexuals. However, this has not been rigorously tested.
To address these questions, our current interests are to (1) understand the genetic mechanism for the origin of oblligate asexuality and (2) genetic consequences of aborting meiosis using obligately asexual Daphnia pulex. Our previous work has clearly demonstrated that the introgression of a set of alleles originated in a closely related species Daphnia pulicaria is perfectly associated with obligate asexuality, opening up an excellent opportunity to understand how these introgressed alleles confer asexuality, i.e., modifying meiosis to a chromosomally non-reduced cell division in germline cells. Furthermore, using mutation-accumulation experiments we showed that large-scale (e.g., tens of thousands basepairs) deletions occurs at extremely high frequency in asexual Daphnia genome, thousands of times higher than point mutations. This finding sets up a neat platform for our future investigation on the genetic consequences of asexuality. We are currently using a variety of epigenetic (e.g., Chip-seq), DNA and RNA sequencing, and mutation-accumulation to further explore these issues.
Despite the common occurrence of meiosis in most sexual eukaryotes, we lack a refined understanding of the evolutionary and/or genetic factors that regulate the occurrence of meiotic crossover and gene conversion events in sexual reproduction. Previous work based on a few classic model organisms (i.e., Saccharomyces cerevisiae, Drosophila melanogaster, mice, and humans) shows that recombination activities are highly clustered in genomes. With the exception of the gene prdm9 (PR domain zinc finger protein 9) in humans and mice, no genetic elements have been determined as a major determinant of recombination hotspots. The narrow taxonomic scope of previous studies also prevents a broader picture of the evolution of recombination patterns. To expand our understanding of the genetic regulation of meiotic recombination, our lab has developed single-sperm whole-genome sequencing in Daphnia to directly examine recombination events in gametes. This effort has resulted in a high-density genetic map of Daphnia pulex using 104 single sperm, which anchors 90% of the resolved genomic sequences in Daphnia genome into chromosome. These data will also generate novel insight into the genomic occurrence of recombination events in Daphnia genome, which we are continuing to work on. The sperm sequencing technique can be easily applied to other Cladoceran speceis that are closely related to Daphnia, offering a rich source of data for understanding the evolution of recombination. Lastly, given that cladocerans probably occupy diverse aquatic habitats ranging from ephemeral ponds, stratified lakes, and brackish/marine water, we will further explore how different environments modulate the rate of recombination within species.
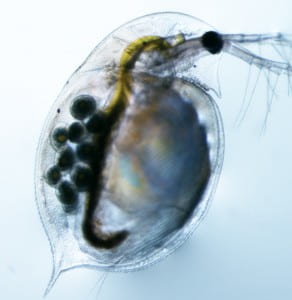
Our research uses the freshwater microcrustacean Daphnia pulex as a model. Daphnia can reproduce clonally for indefinite periods, with periods of environmental stress triggering male production and sexual reproduction. What makes D. pulex unique is the presence of obligate parthenogenetic lineages that have abandoned sexual reproduction, providing a great system for examining the genetic consequences of asexuality. Moreover, D. pulex occupies more diverse environments than perhaps any other aquatic species (from ephemeral woodland ponds to large pristine lakes to high-UV alpine and dune environments to habitats modified dramatically by chemical pollutants), allowing us to test the hypotheses concerning how environmental factors regulate the frequency of sex and recombination in natural populations. The Daphnia system also has the advantage of a large battery of available genomic tools (e.g., multiple reference assemblies, high-resolution genetic map, RNAi). Although D. pulex is currently our primary model organism, we are expanding our lineup of favorite organisms to study their genomic dynamics!